茚达特罗
化合物
(重定向自C24H28N2O3)
茚达特罗(INN:indacaterol)是诺华公司开发的超长效β-肾上腺素受体激动剂。[5]与相关药物福莫特罗和沙美特罗不同,它每天只需服用一次。[6]该药物仅被许可用于治疗慢性阻塞性肺病(迄今为止缺乏哮喘患者的长期数据)。它可通过干粉吸入器以气雾剂形式输送。
 | |
 | |
| 临床资料 | |
|---|---|
| 商品名 | Onbrez Breezhaler、Arcapta Neohaler |
| AHFS/Drugs.com | 国际药品名称 |
| 核准状况 |
|
| 怀孕分级 |
|
| 给药途径 | 吸入 |
| ATC码 | |
| 法律规范状态 | |
| 法律规范 |
|
| 识别信息 | |
| |
| CAS号 | 312753-06-3 |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.218.577 |
| 化学信息 | |
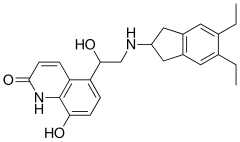
| 化学式 | C24H28N2O3 |
| 摩尔质量 | 392.50 g·mol−1 |
| 3D模型(JSmol) | |
| |
| |
医疗用途
编辑考科蓝合作组织的一项综述发现,茚达特罗在改善慢性阻塞性肺病患者的肺功能方面效果至少与每日两次长效β2受体激动剂相当。[7]
历史
编辑它于2009年11月30日获得欧洲药品管理局批准,商品名为Onbrez Breezhaler,[8]。它也于2011年7月1日获得美国食品和药物管理局批准,商品名为Arcapta Neohaler。[9][10]2016年,诺华将Arcapta Neohaler的美国商业权授权给Sunovion制药。[11]
参考资料
编辑- ^ Arcapta Neohaler (indacaterol) inhalation powder Initial U.S. Approval: 2011. DailyMed. 1 April 2020 [14 June 2021]. (原始内容存档于2021-06-15).
- ^ Onbrez Breezhaler EPAR. European Medicines Agency (EMA). 17 September 2018 [20 January 2021]. (原始内容存档于2021-01-28).
- ^ Oslif Breezhaler EPAR. European Medicines Agency (EMA). 17 September 2018 [20 January 2021]. (原始内容存档于2021-01-28).
- ^ Hirobriz Breezhaler EPAR. European Medicines Agency (EMA). 17 September 2018 [20 January 2021]. (原始内容存档于2021-01-28).
- ^ Cazzola M, Matera MG, Lötvall J. Ultra long-acting beta 2-agonists in development for asthma and chronic obstructive pulmonary disease. Expert Opin Investig Drugs. July 2005, 14 (7): 775–83. PMID 16022567. S2CID 11930383. doi:10.1517/13543784.14.7.775.
- ^ Beeh KM, Derom E, Kanniess F, Cameron R, Higgins M, van As A. Indacaterol, a novel inhaled beta2-agonist, provides sustained 24-h bronchodilation in asthma. Eur. Respir. J. May 2007, 29 (5): 871–8. PMID 17251236. doi:10.1183/09031936.00060006 .
- ^ Geake, James B. Indacaterol, a once-daily beta2-agonist, versus twice-daily beta2-agonists or placebo for chronic obstructive pulmonary disease. Reviews. 2015, 1 (3): CD010139. PMC 6464646 . PMID 25575340. doi:10.1002/14651858.CD010139.pub2.
- ^ European Public Assessment Report for Onbrez Breezhaler 互联网档案馆的存档,存档日期2010-01-16.
- ^ FDA approves Arcapta Neohaler to treat chronic obstructive pulmonary disease (新闻稿). U.S. Food and Drug Administration. 2011-07-01 [2011-07-02]. (原始内容存档于2011-07-03).
- ^ Drug Approval Package: Arcapta Neohaler (indacaterol maleate) NDA #022383. U.S. Food and Drug Administration. 13 August 2013 [14 June 2021]. (原始内容存档于2021-04-07).
- ^ Faulkner, Sarah. Sunovion, Novartis ink licensing deal for inhaled COPD drugs. Drug Delivery Business. 22 December 2016 [2024-05-23]. (原始内容存档于2021-10-31).