黃芩苷
化合物
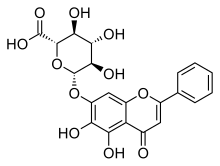
黃芩苷(英語:Baicalin)是一種黃酮類化合物,是黃芩素(Baicalein)與葡萄糖醛酸形成的糖苷,提取自黃芩(Scutellaria baicalensis)[1]、盔狀黃芩(Scutellaria galericulata)[2]等植物。
| 黃芩苷 | |
|---|---|
| |
| IUPAC名 (2S,3S,4S,5R,6S)-6-[(5,6-Dihydroxy-4-oxo-2-phenyl-4H-1-benzopyran-7-yl)oxy]-3,4,5-trihydroxyoxane-2-carboxylic acid | |
| 別名 | 黃芩素 7-O-葡萄糖醛酸 |
| 識別 | |
| CAS號 | 21967-41-9 |
| PubChem | 64982 |
| ChemSpider | 58507 |
| SMILES |
|
| InChI |
|
| InChIKey | IKIIZLYTISPENI-ZFORQUDYBD |
| Beilstein | 70480 |
| ChEBI | 2981 |
| KEGG | C10025 |
| 性質 | |
| 化學式 | C21H18O11 |
| 摩爾質量 | 446.36 g·mol−1 |
| 熔點 | 202—205 °C(396—401 °F;475—478 K) |
| 危險性 | |
GHS危險性符號
| |
| GHS提示詞 | Warning |
| H-術語 | H315, H319, H335 |
| P-術語 | P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405 |
| 若非註明,所有數據均出自標準狀態(25 ℃,100 kPa)下。 | |
藥用價值
編輯黃芩苷及其糖苷配基黃芩素都是苯二氮䓬受體的正別構調節劑。[4][5][6]小鼠實驗表明黃芩苷是抗焦慮藥,但不是鎮靜劑或肌肉鬆弛劑。[7][8]黃芩和盔狀黃芩的抗焦慮作用可能源自黃芩苷及其它黃酮類化合物。[9][10]
參考文獻
編輯- ^ 1.0 1.1 Su, Hai-xia; Yao, Sheng; Zhao, Wen-Feng; Li, Min-jun; Liu, Jia; Shang, Wei-Juan; Xie, Hang; Ke, Chang-Qiang; Hu, Hang-Chen; Gao, Mei-na; Yu, Kun-Qian; Liu, Hong; Shen, Jing-Shan; Tang, Wei; Zhang, Lei-ke; Xiao, Geng-fu; Ni, Li; Wang, Dao-wen; Zuo, Jian-Ping; Jiang, Hua-Liang; Bai, Fang; Wu, Yan; Ye, Yang; Xu, Ye-Chun. Anti-SARS-CoV-2 activities in vitro of Shuanghuanglian preparations and bioactive ingredients. Acta Pharmacologica Sinica. 2020, 41 (9): 1167–1177. PMC 7393338 . PMID 32737471. doi:10.1038/s41401-020-0483-6.
- ^ P.H. and Horhammer, L., Hager's Handbuch der Pharmazeutischen Praxis, Vols. 2-6, Springer-Verlag, Berlin, 1969-1979
- ^ Taira, Zenei; Yabe, Kazunori; Hamaguchi, Yuka; Hirayama, Kensuke; Kishimoto, Makoto; Ishida, Shiro; Ueda, Yukari. Effects of Sho-saiko-to extract and its components, Baicalin, baicalein, glycyrrhizin and glycyrrhetic acid, on pharmacokinetic behavior of salicylamide in carbon tetrachloride intoxicated rats. Food and Chemical Toxicology (Elsevier BV). 2004, 42 (5): 803–807. ISSN 0278-6915. doi:10.1016/j.fct.2003.12.017.
- ^ Wang H, Hui KM, Xu S, Chen Y, Wong JT, Xue H. Two flavones from Scutellaria baicalensis Georgi and their binding affinities to the benzodiazepine site of the GABAA receptor complex. Pharmazie. 2002, 57 (12): 857–8. PMID 12561253.
- ^ Hui KM, Wang XH, Xue H. Interaction of flavones from the roots of Scutellaria baicalensis with the benzodiazepine site. Planta Med. 2000, 66 (1): 91–3. PMID 10705749. S2CID 260249283. doi:10.1055/s-0029-1243121.
- ^ Edwin Lowell Cooper; Nobuo Yamaguchi. Complementary and Alternative Approaches to Biomedicine. Springer Science & Business Media. 1 January 2004: 188–. ISBN 978-0-306-48288-5.
- ^ Xu Z, Wang F, Tsang SY, Ho KH, Zheng H, Yuen CT, Chow CY, Xue H. Anxiolytic-Like Effect of baicalin and its additivity with other anxiolytics. Planta Med. 2006, 72 (2): 189–92. PMID 16491459. S2CID 2398014. doi:10.1055/s-2005-873193.
- ^ Liao JF, Hung WY, Chen CF. Anxiolytic-like effects of baicalein and baicalin in the Vogel conflict test in mice. Eur. J. Pharmacol. 2003, 464 (2–3): 141–6. PMID 12620506. doi:10.1016/s0014-2999(03)01422-5.
- ^ Awad R, Arnason JT, Trudeau V, Bergeron C, Budzinski JW, Foster BC, Merali Z. Phytochemical and biological analysis of skullcap (Scutellaria lateriflora L.): a medicinal plant with anxiolytic properties. Phytomedicine. 2003, 10 (8): 640–9. PMID 14692724. doi:10.1078/0944-7113-00374.
- ^ Stefanie Schwartz. Psychoactive Herbs in Veterinary Behavior Medicine. John Wiley & Sons. 9 January 2008: 139–. ISBN 978-0-470-34434-7.
- ^ Tarragó, T; Kichik, N; Claasen, B; Prades, R; Teixidó, M; Giralt, E. Baicalin, a prodrug able to reach the CNS, is a prolyl oligopeptidase inhibitor. Bioorganic & Medicinal Chemistry. 2008, 16 (15): 7516–24. PMID 18650094. doi:10.1016/j.bmc.2008.04.067.
- ^ Takahashi H, Chen MC, Pham H, Angst E, King JC, Park J, Brovman EY, Ishiguro H, Harris DM, Reber HA, Hines OJ, Gukovskaya AS, Go VL, Eibl G. Baicalein, a component of Scutellaria baicalensis, induces apoptosis by Mcl-1 down-regulation in human pancreatic cancer cells. Biochim Biophys Acta. 2011, 1813 (8): 1465–1474. PMC 3123440 . PMID 21596068. doi:10.1016/j.bbamcr.2011.05.003.