依布硒
化合物
依布硒(英語:Ebselen、也称为PZ 51、DR3305和SPI-1005)是一种合成的有机硒药物分子,具有抗炎、抗氧化和细胞保护活性。它充当谷胱甘肽过氧化物酶的模拟物,也可以与过氧亚硝酸盐反应。[1]正在被研究作为再灌注损伤和中风、[2][3]听觉损失和耳鸣[4][5]和双向情绪障碍症[6][7]的可能治疗药物。
| 依布硒 | |
|---|---|
| |

| |

| |
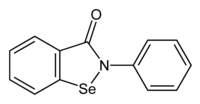
| IUPAC名 2-Phenyl-1,2-benzoselenazol-3(2H)-one | |
| 识别 | |
| CAS号 | 60940-34-3 |
| PubChem | 3194 |
| ChemSpider | 3082 |
| SMILES |
|
| InChI |
|
| InChIKey | DYEFUKCXAQOFHX-UHFFFAOYAZ |
| ChEBI | 77543 |
| 性质 | |
| 化学式 | C13H9NOSe |
| 摩尔质量 | 274.17666 g·mol⁻¹ |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
此外,依布硒也可以有效地对抗艰难拟梭菌感染[8]且它也被证明拥有对抗烟曲霉的抗真菌活性。[9]
依布硒是过氧化氢和氢过氧化物(包括膜结合磷脂和胆固醇酯氢过氧化物)的有效清除剂。几种依布硒类似物已显示在硫醇存在下清除过氧化氢。[10]
可能的抗COVID-19活性
编辑初步研究表明,依布硒在基于细胞的测定中表现出对COVID-19的抑制活性。[11][12][13]该效应归因于通过与活性中心的半胱氨酸(Cys-145)的硫醇基团形成共价键对主要蛋白酶的不可逆抑制。[11]
合成
编辑通常,依布硒的特征支架,苯并异硒唑酮环系统的合成,可以通过伯胺(RNH2)与2-(氯硒)苯甲酰氯(路线一)的反应、[14]苯甲酰苯胺的邻位锂化和氧化环化来实现(路线二)由溴化铜(CuBr2)介导,[15]或通过高效的铜催化的邻卤代苯甲酰胺的硒化/杂环化,这是Kumar等人开发的一种方法[16](路线三)。
参考文献
编辑- ^ Schewe T. Molecular actions of ebselen--an antiinflammatory antioxidant. General Pharmacology. October 1995, 26 (6): 1153–69. PMID 7590103. doi:10.1016/0306-3623(95)00003-J.
- ^ Parnham M, Sies H. Ebselen: prospective therapy for cerebral ischaemia. Expert Opinion on Investigational Drugs. March 2000, 9 (3): 607–19. PMID 11060699. S2CID 42599736. doi:10.1517/13543784.9.3.607.
- ^ Yamaguchi T, Sano K, Takakura K, Saito I, Shinohara Y, Asano T, Yasuhara H. Ebselen in acute ischemic stroke: a placebo-controlled, double-blind clinical trial. Ebselen Study Group. Stroke. January 1998, 29 (1): 12–7. PMID 9445321. doi:10.1161/01.STR.29.1.12 .
- ^ Kil J, Pierce C, Tran H, Gu R, Lynch ED. Ebselen treatment reduces noise induced hearing loss via the mimicry and induction of glutathione peroxidase. Hearing Research. April 2007, 226 (1–2): 44–51. PMID 17030476. S2CID 39896274. doi:10.1016/j.heares.2006.08.006.
- ^ Kil, Jonathan; Harruff, E. Emily; Longenecker, Ryan J. Development of ebselen for the treatment of sensorineural hearing loss and tinnitus. Hearing Research (Elsevier BV). 2022, 413: 108209. ISSN 0378-5955. PMID 33678494. S2CID 231956488. doi:10.1016/j.heares.2021.108209.
- ^ Singh N, Halliday AC, Thomas JM, Kuznetsova OV, Baldwin R, Woon EC, et al. A safe lithium mimetic for bipolar disorder. Nature Communications. 2013, 4: 1332. Bibcode:2013NatCo...4.1332S. PMC 3605789 . PMID 23299882. doi:10.1038/ncomms2320.
- ^ New drug for bipolar disorder may offer fewer side effects. University of Oxford Latest News. 2013-01-08 [12 January 2013]. (原始内容存档于2020-08-05).
- ^ Drug disarms deadly C. difficile bacteria without destroying healthy gut flora. Medical Express. [2022-10-22]. (原始内容存档于2020-09-23).
- ^ Marshall AC, Kidd SE, Lamont-Friedrich SJ, Arentz G, Hoffmann P, Coad BR, Bruning JB. Structure, Mechanism, and Inhibition of Aspergillus fumigatus Thioredoxin Reductase. Antimicrobial Agents and Chemotherapy. March 2019, 63 (3): e02281–18. PMC 6395915 . PMID 30642940. doi:10.1128/AAC.02281-18 .
- ^ Satheeshkumar K, Mugesh G. Synthesis and antioxidant activity of peptide-based ebselen analogues. Chemistry. April 2011, 17 (17): 4849–57. PMID 21400619. doi:10.1002/chem.201003417.
- ^ 11.0 11.1 Jin Z, Du X, Xu Y, Deng Y, Liu M, Zhao Y, et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature. June 2020, 582 (7811): 289–293. Bibcode:2020Natur.582..289J. PMID 32272481. doi:10.1038/s41586-020-2223-y .
- ^ Weglarz-Tomczak E, Tomczak JM, Talma M, Burda-Grabowska M, Giurg M, Brul S. Identification of ebselen and its analogues as potent covalent inhibitors of papain-like protease from SARS-CoV-2. Scientific Reports. February 2021, 11 (1): 3640. Bibcode:2021NatSR..11.3640W. PMC 7878891 . PMID 33574416. doi:10.1038/s41598-021-83229-6.
- ^ Xiang R, Yu Z, Wang Y, Wang L, Huo S, Li Y, et al. Recent advances in developing small-molecule inhibitors against SARS-CoV-2. Acta Pharmaceutica Sinica. B. July 2021, 12 (4): 1591–1623. PMC 8260826 . PMID 34249607. doi:10.1016/j.apsb.2021.06.016.
- ^ Kamigata N, Iizuka H, Izuoka A, Kobayashi M. Photochemical Reaction of 2-Aryl-1, 2-benzisoselenazol-3 (2 H)-ones.. Bulletin of the Chemical Society of Japan. July 1986, 59 (7): 2179–83. doi:10.1246/bcsj.59.2179 .
- ^ Engman L, Hallberg A. Expedient synthesis of ebselen and related compounds. The Journal of Organic Chemistry. 1989-06-01, 54 (12): 2964–2966. ISSN 0022-3263. doi:10.1021/jo00273a035.
- ^ Balkrishna SJ, Bhakuni BS, Chopra D, Kumar S. Cu-catalyzed efficient synthetic methodology for ebselen and related Se-N heterocycles. Organic Letters. December 2010, 12 (23): 5394–7. PMID 21053969. doi:10.1021/ol102027j.