雷美替胺 編輯
化合物
此條目可參照英語維基百科相應條目來擴充。 (2024年1月26日) |
雷美替胺,商品名柔速瑞(Rozerem),是可治療因難以入睡而失眠的褪黑激素受體激動劑。[2][4]它可減少入睡所需的時間,但臨床益處較少。[5]它是口服藥。[2]
 | |
 | |
| 臨床資料 | |
|---|---|
| 商品名 | Rozerem, others |
| 其他名稱 | TAK-375 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605038 |
| 核准狀況 | |
| 依賴性 | 低[1] |
| 給藥途徑 | 口服 |
| ATC碼 | |
| 法律規範狀態 | |
| 法律規範 | |
| 藥物動力學數據 | |
| 生物利用度 | 1.8%[2] |
| 血漿蛋白結合率 | 82%(大多為白蛋白)[2] |
| 藥物代謝 | 肝(CYP1A2,少部分由CYP2C與CYP3A4代謝)[2] |
| 代謝產物 | M-II(活性代謝產物)[2] |
| 生物半衰期 | 雷美替胺:1–2.6小時[2] M-II:2–5小時[2][3] |
| 排泄途徑 | 尿液84%[2] 糞便4%[2] |
| 識別資訊 | |
| |
| CAS號 | 196597-26-9 |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.215.666 |
| 化學資訊 | |
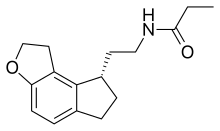
| 化學式 | C16H21NO2 |
| 摩爾質量 | 259.35 g·mol−1 |
| 3D模型(JSmol) | |
| |
| |
雷美替胺的副作用包括昏睡、頭暈、疲勞、噁心、失眠加劇、激素水平改變。[2]它是褪黑激素的結構類似物,也是褪黑激素受體1A和褪黑激素受體1B的選擇性激動劑。[2]雷美替胺的生物半衰期和藥效持續時間都比褪黑激素長。[6]它不是苯二氮䓬類或非苯二氮䓬類藥物,不作用於GABA受體,有獨特的作用機制。[2][7]
參考資料
編輯- ^ Kim, HK; Yang, KI. Melatonin and melatonergic drugs in sleep disorders.. Translational and clinical pharmacology. December 2022, 30 (4): 163–171. PMC 9810491 . PMID 36632077. doi:10.12793/tcp.2022.30.e21 .
- ^ 移至: 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 Rozerem- ramelteon tablet, film coated. DailyMed. 2018-12-28 [2020-04-13]. (原始內容存檔於2021-03-26).
- ^ Karim A, Tolbert D, Cao C. Disposition kinetics and tolerance of escalating single doses of ramelteon, a high-affinity MT1 and MT2 melatonin receptor agonist indicated for treatment of insomnia. Journal of Clinical Pharmacology. February 2006, 46 (2): 140–148. PMID 16432265. S2CID 38171735. doi:10.1177/0091270005283461.
- ^ Neubauer DN. A review of ramelteon in the treatment of sleep disorders. Neuropsychiatric Disease and Treatment. February 2008, 4 (1): 69–79. PMC 2515902 . PMID 18728808. doi:10.2147/ndt.s483 .
- ^ Kuriyama A, Honda M, Hayashino Y. Ramelteon for the treatment of insomnia in adults: a systematic review and meta-analysis. Sleep Medicine. April 2014, 15 (4): 385–392. PMID 24656909. doi:10.1016/j.sleep.2013.11.788.
- ^ Hardeland R, Poeggeler B, Srinivasan V, Trakht I, Pandi-Perumal SR, Cardinali DP. Melatonergic drugs in clinical practice. Arzneimittel-Forschung. 2008, 58 (1): 1–10. PMID 18368944. S2CID 38857779. doi:10.1055/s-0031-1296459.
- ^ Atkin T, Comai S, Gobbi G. Drugs for Insomnia beyond Benzodiazepines: Pharmacology, Clinical Applications, and Discovery. Pharmacological Reviews. April 2018, 70 (2): 197–245. PMID 29487083. S2CID 3578916. doi:10.1124/pr.117.014381 .
- ^ Uchikawa O, Fukatsu K, Tokunoh R, Kawada M, Matsumoto K, Imai Y, et al. Synthesis of a novel series of tricyclic indan derivatives as melatonin receptor agonists. Journal of Medicinal Chemistry. September 2002, 45 (19): 4222–4239. PMID 12213063. doi:10.1021/jm0201159.
- ^ Drug Approval Package: Rozerem (Ramelteon) NDA #021782. U.S. Food and Drug Administration (FDA). 2005-10-20 [2020-04-13]. (原始內容存檔於2021-03-31).